 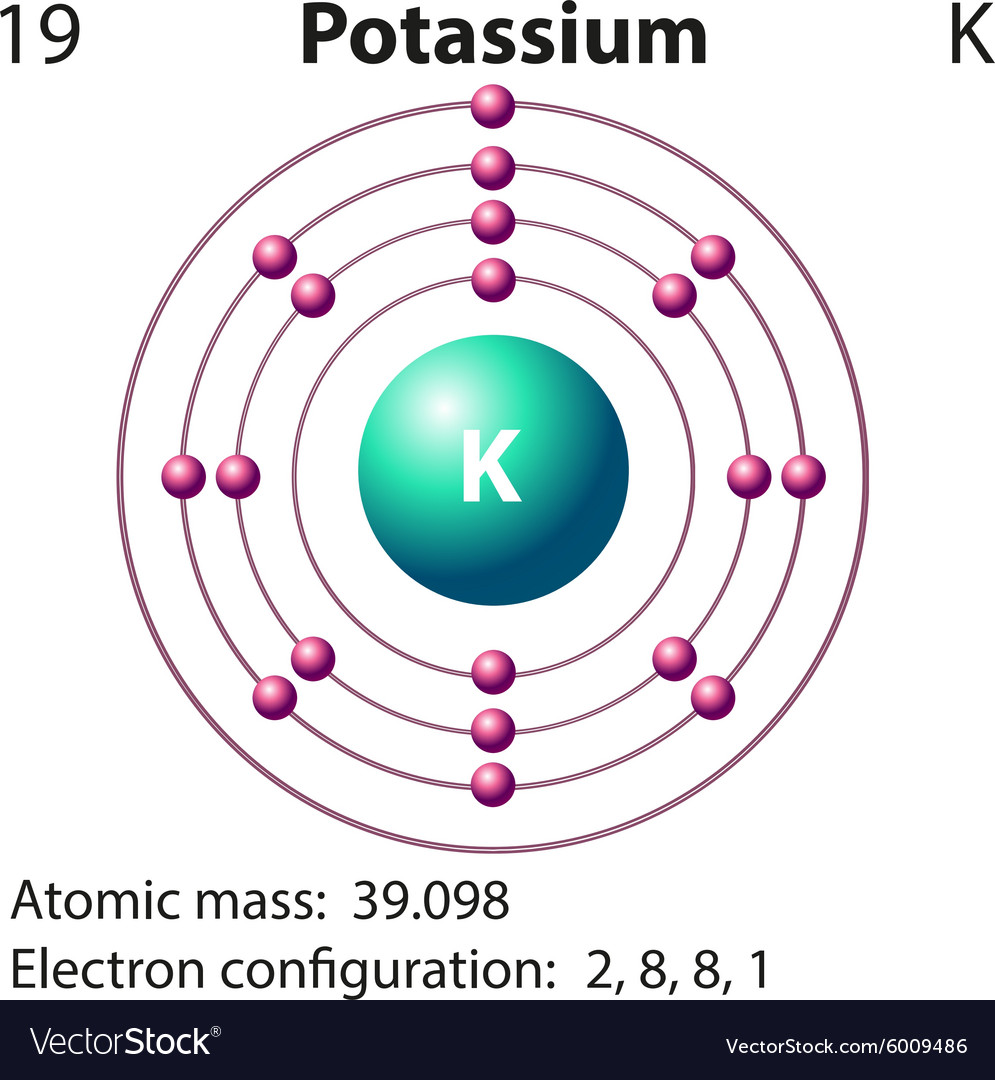
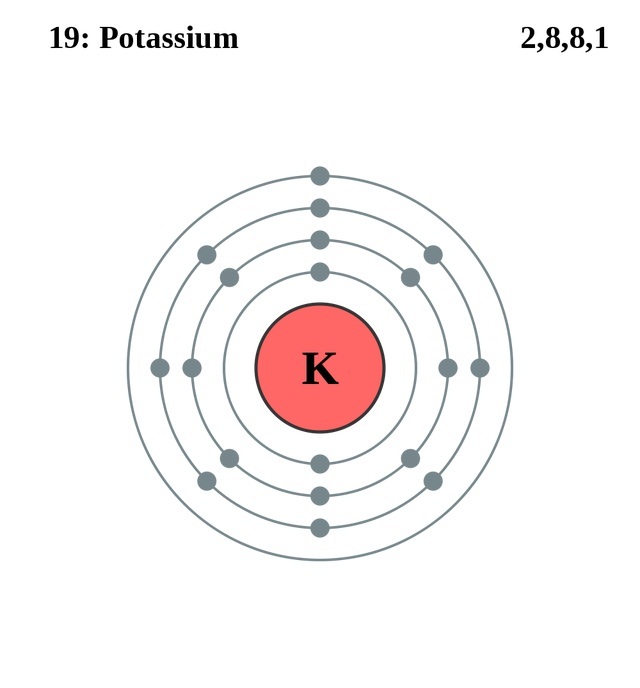
the amount of screening by the inner electrons,. 
Ionization energy is governed by three factors: Notice that first ionization energy decreases down the group. The first ionization energy of an atom is defined as the energy required to remove the most loosely held electron from each of one mole of gaseous atoms, producing one mole of singly charged gaseous ions in other words, it is the energy required for 1 mole of this process:Ī graph showing the first ionization energies of the Group 1 atoms is shown above. Therefore, the atoms increase in size down the group. More layers of electrons take up more space, due to electron-electron repulsion. The only factor affecting the size of the atom is the number of layers of inner electrons which surround the atom. This is true for each of the other atoms in Group 1. This effect is illustrated in the figure below: The positive charge on the nucleus is canceled out by the negative charges of the inner electrons. In each element, the outer electron experiences a net charge of 1 from the nucleus.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
